Diabetes incidence has skyrocketed in recent decades
due to our dietary and life-style choices - i.e., we are too fat, eat bad
foods, and don’t exercise enough. Unfortunately, due to extensive
metabolic, hormonal, body-composition shifts that occur after spinal cord
injury (SCI), people with SCI are especially prone to develop diabetes.
This article discusses various nutritional or botanical approaches that
reduce blood-sugar levels in people with this disease.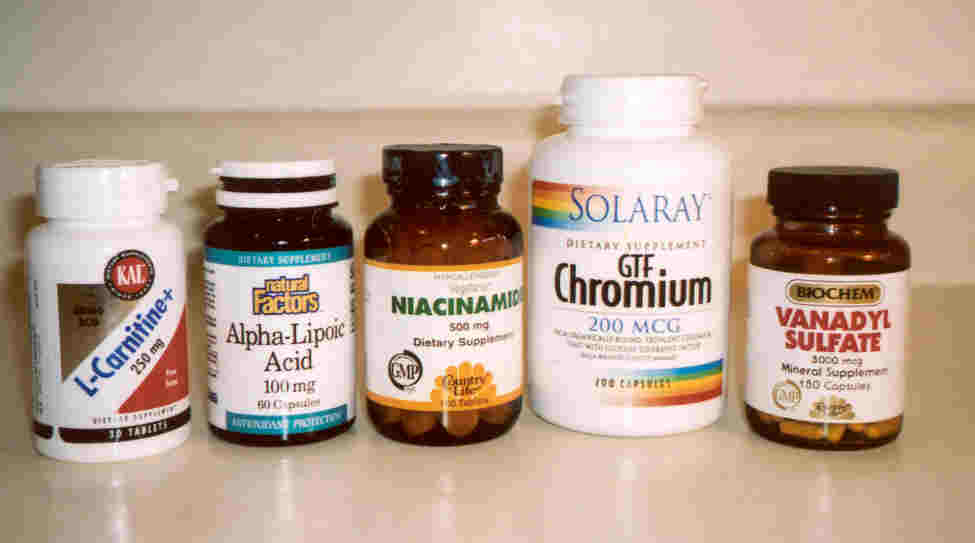
Overview
Diabetes is characterized by the body’s inability to
properly use glucose, our body’s metabolic energy currency. Under healthy
conditions, the hormone insulin controls blood-glucose levels. Produced by
the pancreas, insulin flows through our blood to various tissues where
they bind to cell-surface receptors. This binding initiates a complex
biochemical cascade that culminates in glucose uptake into cells to fuel
metabolic processes. As blood-glucose levels decline due to cellular
uptake, the pancreas shuts down insulin production to prevent hypoglycemia
(low blood sugar) and, in turn, the liver, the body’s nutrient-processing
organ, starts releasing glucose back into the blood.
Although distinctions are more blurred than once
thought, in type-1 (“juvenile”) diabetes, the pancreas produces inadequate
insulin to promote glucose uptake into cells. In contrast, in type-2
(“adult-onset diabetes”), cells become less responsive to the insulin that
is still produced.
Through not clearly defined physiological mechanisms,
persistently high blood glucose may eventually lead to complications such
as kidney disease, neuropathy (nerve disease), and eye problems.
Diabetes and SCI
Dr. William Bauman and colleagues at the Bronx VA
Medical Center have shown that SCI predisposes one to diabetes. For
example, their research indicates that 1) only 44% of veterans with SCI
have normal glucose metabolism compared to 82% without SCI and 2)
higher-level injuries further predispose individuals to diabetes.
Nutrient Density & SCI
Before our sedentary modern age, mankind had high
caloric needs due to more intense manual labor. If 4,000 calories/day were
needed to fuel such labor, one was much more likely to absorb trace
nutrients compared to today’s office worker who gains weight from 1,500
calories/day of nutrient-depleted, processed foods.
This concept is especially relevant to SCI. For
example, by comparing identical twins with and without SCI, Bauman has
shown that caloric needs are greatly reduced after injury. Such a
reduction automatically results in the uptake of fewer trace nutrients in
a weight-maintaining diet.
Another factor compromising nutrient uptake is
extensive antibiotic use - a life-saving cornerstone of SCI medicine. A
Science magazine article (Gilmore & Ferretti, March 28, 2003) noted:
"Not only does the highly evolved gut flora community extend the
processing of undigested food to the benefit of the host, but it also
contributes to host defense by limiting colonization of the
gastrointestinal tract by pathogens." More simply stated, there are
hundreds of bacterial species within our gut that are essential for proper
digestion. Every time you use an antibiotic, assistive bacteria are
killed, in turn, lessening the uptake of nutrients already inherently
compromised by SCI.
The gist of this discussion is people with SCI need
to make every calorie count, because the adverse ramifications of eating
nutrient-depleted food will be greatly magnified for them.
Given these considerations, the effectiveness of
supplementing your diet with various nutrients to reduce blood-glucose
levels depends upon how much of the nutrients you already absorb from your
diet. If your diet already contains an abundance of a nutrient, a
supplement isn’t going to help; but if it is lacking, it may provide
significant benefit.
Many of the foods, supplements, or botanical agents
discussed below are supported by scientific studies (including, in some
cases, rigorous double-blind clinical studies) and are readily available
from nutritional, health-food or ethnic-grocery stores, or Internet
sources.
Mineral Supplements
Trace amounts of chromium are essential for
proper carbohydrate metabolism. Depending upon dietary intake, studies
suggest that chromium supplementation lowers blood-glucose levels by
increasing cell-surface insulin receptors and, in turn, glucose transport
into cells. Traditional food sources of chromium include beer, brewer’s
yeast, brown rice, cheese, meat, and whole grains.
Before insulin, vanadium was a traditional
French diabetes remedy. Animal and human studies indicate that vanadium,
like chromium, facilitates glucose uptake and metabolism by enhancing
insulin-receptor expression. Vanadium is found in fish, vegetable oils,
and olives. Excess vanadium supplementation should be avoided.
Diabetics often have low levels of magnesium,
a mineral required for many physiological functions. Individuals with the
least amount of magnesium in their blood have twice the diabetes incidence
as those with the highest levels. Scientists have shown that when
magnesium-deficient patients were given supplements, their responsiveness
to insulin and glucose metabolism improved. Fish, meat, seafood and nuts
are rich magnesium sources.
Other Supplements
The antioxidant alpha-lipoic acid is a common
German treatment for diabetic neuropathy (antioxidants protect our bodies
from cell-damaging, free radicals). Basically, with such neuropathy,
persistent high blood glucose generates oxidative stress that compromises
blood flow to nerves, damaging signal-conducting axons. Over time, this
damage may lead to, for example, foot problems requiring amputation.
Through its antioxidant properties, alpha-lipoic acid enhances nerve
health, lessening neuropathy. Red meat is a good source of alpha-lipoic
acid.
The amino acid carnitine plays a key role in
fat and glucose metabolism. Evidence suggests it lowers glucose levels
through increasing glucose metabolism (i.e., burning it off) and creating
glucose polymers called glycogen (i.e., putting it in storage).
Double-blind clinical studies indicate that carnitine helps diabetic
neuropathy by enhancing neuronal conduction and reducing pain. Beef is the
richest source of carnitine.
The vitamin nicotinamide (a biochemical
derivative of niacin) lowers blood-glucose levels by enhancing the
pancreas’ insulin-producing ability. For example, blood-glucose metabolism
was improved in type-2 diabetics who had lost responsiveness to commonly
prescribed sulfonylurea drugs (sulfonylureas make the pancreas release
more insulin) after consuming nicotinamide for six months. Meat, fish and
poultry are good sources of this vitamin.
Foods and Botanical Medicines
Before insulin was developed, diabetes was treated
throughout the world with a multitude of traditional plant remedies:
Ginseng is a traditional cure-all that
restores a more health-promoting balance or homeostasis to the body.
Studies show that diabetics who consume ginseng (American species) before
an oral glucose challenge had lower blood-glucose levels. With such a
challenge, fasting patients consume glucose, and then its elimination from
the blood is followed over time. The results suggest that consuming
ginseng before a meal could lower blood-sugar levels after the meal (i.e.,
postprandial).
Unripe bitter melon, a cucumber-shaped fruit,
is a traditional Asian diabetes remedy. In one study, diabetics who drank
juiced bitter-melon had lower fasting and postprandial blood-glucose
levels. Bitter melon includes physiologically active agents that affect
glucose metabolism, including an insulin-resembling molecule.
Related to milkweed, gymnema is an Ayurvedic
(India’s ancient medicine) remedy for diabetes. Diabetics who consumed
gymnema for 18-20 months had lower blood-sugar levels, and a significant
number were able to discontinue conventional oral medications. In rats,
gymnema doubled pancreatic, insulin-producing beta cells.
Fenugreek, a common Indian and Middle-Eastern
spice, possesses anti-diabetic properties. In one study, diabetics who
consumed fenugreek seed powder at lunch and dinner for six months had
substantially lower fasting blood glucose and demonstrated improved
glucose-tolerance test results. Because fenugreek is somewhat bitter, it
can also be taken in capsules available at health-food stores.
The pads and fruits of the prickly pear cactus or
nopal are Hispanic foods that are also traditional anti-diabetic
remedies. Several studies demonstrate the ability of nopal, which contains
substances that increase insulin sensitivity, to reduce blood-glucose
levels in diabetics. Nopal can be consumed raw, cooked or juiced.
Another desert plant with anti-diabetic properties is
aloe vera. Best known as a topical remedy for burns and wounds, it
is also a traditional Arabian Peninsula diabetes remedy. Studies suggest
that diabetics who consume aloe gel daily over time can substantially
lower blood-glucose levels.
Eating nuts and nut butters reduces diabetes
risk, probably due to their fiber, healthy fats and magnesium. In a
nutshell, Harvard investigators followed 83,000 women and found that those
who ate an ounce of nuts or a tablespoon of peanut butter five times a
week or more reduced their risk of diabetes by nearly 30%.
Other potential anti-diabetes foods include 1)
noni juice (Morinda citrifolia), a traditional Polynesian cure-all for
many disorders; 2) cinnamon; 3) jambul, a plant related to
cloves and another Ayurvedic remedy; and 4) stevia, a popular sugar
substitute isolated from a South-American shrub.
Fiber
Evidence suggests that diabetes risk can be lowered
by consuming dietary fibers that slow carbohydrate absorption, including
glucomannan, a traditional Japanese food isolated from the konjac
root (related to yams); and psyllium, the laxative Metamucil’s
active ingredient.
Dietary Vices
Coffee addicts rejoice! Validating previous
studies, Harvard investigators followed over 126,000 subjects up to 18
years and found that men who drank over six cups (i.e., 48 oz.) of coffee
daily reduced their diabetes risk by over 50% (30% for women). The effect
faded away with less than three cups per day. Recent research suggests
that coffee contains chemicals that attenuate intestinal glucose
absorption.
In addition, moderate alcohol consumption
lowers diabetes risk, as it does for heart disease. For example, one study
evaluated diabetes incidence in 23,000 Finnish twins. Results indicated
that the twin who drank moderately but not heavily had half the diabetes
risk as the twin who abstained or drank low amounts of alcohol. Among
other mechanisms, moderate alcohol consumption may enhance insulin
sensitivity.
It goes without saying, however, that these
diabetes-related benefits could be offset by other adverse effects.
Native-American Herbs
Native-American medicine emphasizes healing herbs
(see October 2004 PN), including many that target diabetes.
According to American Indian Healing Arts (1999), anti-diabetic
herbs include devil’s club, barberry, uva ursi, Cananda and daisy
fleabane, alum root, joe-pye weed, red trillium, wild ginger, clintonia,
bugleweed, and flowering spurge. Extensive information on each of these
herbs can be obtained through a Google Internet search (www.google.com).
Fats
Fat and carbohydrate metabolism are intimately
connected, explaining why there are fat vegetarians and why people lose
body fat on low-carbohydrate diets. As such, issues involving fat
consumption and metabolism are of paramount importance in diabetes.
One fat that is especially important is linolenic
acid, an essential fatty acid that we must consume from our diet. Once
consumed, we convert it to gamma-linolenic acid, an important
precursor to prostaglandin hormones that regulate blood flow. This
conversion is compromised in diabetics, which, as a result, promotes
diabetic neuropathy. Studies suggest that gamma-linolenic acid
supplementation reduces such neuropathy. Primrose, borage, and black
current oils are rich sources of this fatty acid.
One intriguing theory attributes diabetes development
to the chronic consumption of bad fats and, in turn, suggests that it can
be cured by consuming good fats (see e.g.,
www.healingmatters.com). This theory suggests that diabetes was
relatively rare until the introduction of engineered fats into our food
supply. Over time, these fats became a pervasive component of our diet and
increasingly replaced the more natural fats that our bodies had
physiological evolved to use over eons.
If sufficient amounts of unnatural fats intercalate
into the cell membrane, membrane viscosity increases, in turn,
compromising the ability of embedded insulin receptors to promote glucose
uptake. It’s like starting your car when it is minus 25o F and
your engine still contains last summer’s heavyweight oil. Turnover (i.e.,
glucose uptake) won’t be that efficient.
Conversely, if a diet is shifted to one that
emphasizes more physiologically-compatible fats, the bad fats will be
eventually displaced from the membrane, once again enhancing receptor
function.