|
Restoration of Bowel and Bladder Function in Patients with Paraplegia
by Vascularized Intercostal Nerve Transfer to Sacral Nerve Roots with
Selected Interfascicular Anastomosis |
|
|
ZHANG Shaocheng,
M.D.1, JOHNSTON, Laurance Ph.D.2, HU Yuhua, M.D.1,
MA Yuhai, M.D.1, ZHANG Zhenwei, M.D.1, ZHANG
Chuanshen, M.D.1, and DANG Ruishan, M.D.1
1Department
of Orthopedics, Changhai Hospital, Shanghai, China; 2Grantee,
Paralyzed Veterans of America, USA |
|
ABSTRACT:
Objective:
To restore bowel and bladder function in
chronically injured patients with paraplegia.
Methods:
Two normal vascularized intercoastal nerves above the spinal cord injury
site were harvested by cutting in at their distal ends at the
midclavicular line and separating the proximal ends from the levatores
costarum. The nerves were then transferred to the vertebral canal through
a submuscular tunnel. A sural nerve segment that had been harvested and
sheared into two segments was sutured to the intercostal nerves by
epiperineurial neurorrhaphy and then to the sacral 2-4 nerve roots by
interfascicular neurorrhaphy. Thirty patients were postoperatively
followed for 2-11 years (average 5 years), of which 1) 26 (87%) recovered
partial defecation and urination sensation, 2) 23 (77%) regained the
micturition reflex and uriesthesis; 3) 19 (63%) recovered partial function
of the detrusor and sphincter urethra muscles; 4) 20 (67%) recovered
partial defecation function and sphincter contraction; and 5) 8 (27%)
regained the previous two functions (i.e., 3 & 4).
Conclusion:
Because this surgical intervention creates
an intercostal-sural nerve bridge that bypasses the injury site,
significant bowel and bladder function can be restored in paraplegic
patients with chronic spinal cord injuries.
INTRODUCTION:
Recently, Zhang et al.1
reported the restoration of stepping-forward and ambulatory function in
patients with paraplegia by rerouting vascularized intercostal nerves to
lumbar nerve roots using interfascicular anastomosis. This article
discusses a similar nerve-transfer procedure to restore bowel and bladder
function in individuals with paraplegia.
In spite of society’s frequent focus on
walking, people with SCI consistently indicate that restoration of bowel
and bladder function is their foremost priority, in part, because of the
personal independence and opportunities for societal integration such
restoration can provide. This article describes surgical procedures that
help restore these high-priority functions. Specifically, since 1990, we
have transferred vascularized intercostal nerves connected to an
intervening sural nerve segment to the S2-4 nerve roots with
selected interfascicular anastomosis to restore significant bowel and
bladder function in patients with paraplegia.
MATERIALS AND METHODS:
Subjects:
Nineteen subjects were male and 11 female.
Their age ranged from 19 to 46, averaging 31years. Seventeen cases were
traumatically injured in the T9-11 level and 13 cases at the T12
- L2 level. Eighteen, 4, 5, and 3 cases, respectively,
had been injured by motor vehicle accidents, falls from high places,
falling objects, and firearms. The time between injury and surgery ranged
from 6 to 36 months, averaging 18 months. All subjects had undergone
vertebral lamina decompression and internal fixation, 24 of whom had an
additional operation to remove the fixation.
Surgical Procedures:
The surgical intervention involves three key components: 1) detaching from
above the injury site intercostal nerves which are still connected to the
spinal cord, 2) linking their distal ends to a sural nerve segment
isolated fr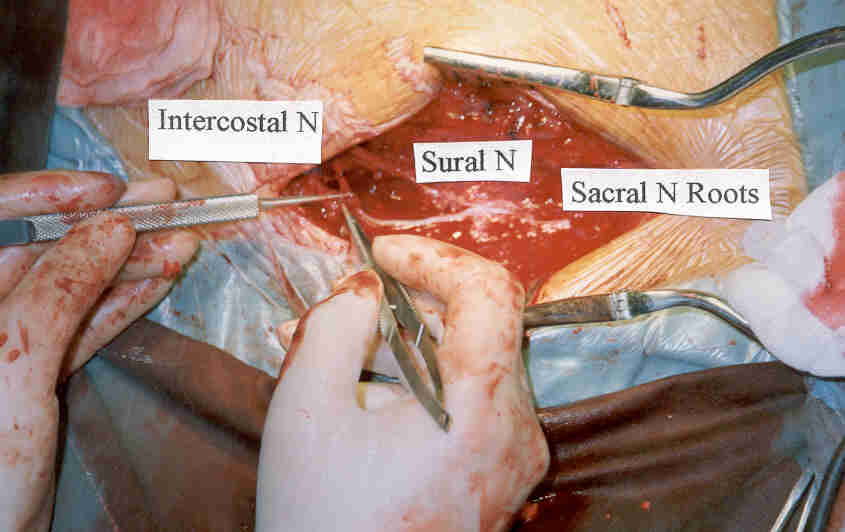 om
the calf, and 3) attaching this combined nerve bridge to the sacral nerve
roots below the injury site. Although several procedural variations have
been used, a representative one is as follows. om
the calf, and 3) attaching this combined nerve bridge to the sacral nerve
roots below the injury site. Although several procedural variations have
been used, a representative one is as follows.
Placed in the lateral position with the
operating side upward, patients were subjected to tracheal intubation with
general anesthesia. Two normal intercostal nerves were chosen for the
procedure, avoiding the selection of lower nerves that may result in the
post-surgical ascension of the paraplegia level (especially sensation).
Along the indicated intercostal space, an incision was made from the
sacrospinal muscle to the midclavicular line. The selected intercostal
nerves were separated from the abdominal muscle, cutoff at the distal
nipple line, and their severed blood vessels ligated. The intact blood
vessels’ proximal ends were moved freely to the edge of the levator
muscle. A channel was created between the vertebral canal, and a lateral
thoracic incision made under the sacrospinal muscle.
Depending on whether the reconnection to
the sacral nerve roots was to be done in the intradural or extradural
space, a median incision was made posterior to either the T12-L2
(intradural) or L5 –S2 (extradural) level. The
old operative scar was removed, and, if necessary, further decompression
of the vertebral lamina was carried out. The S1-2 vertebral
laminas and nerve root tube were cut open with a bone rongeur to expose
the starting ends of the S2-4 nerve roots. If the dura mater
was not injured, the segment was incised to specifically expose the S2-4
nerve roots.
A homolateral sural nerve segment twice
the length of the distance from the intercostal nerve to the sacral nerve
roots was obtained. Because each intercostal nerve has a sensory and motor
branch, one end of the sural nerve segment was divided to create a
Y-shaped segment. The segment’s proximal ends were extra-fascicularly
sutured with the intercostal nerve, and the distal end, after passing
through the submuscular channel, was branched and sutured with either the
S2-3 or S3-4 nerve roots.
In order not to damage the lower reflex
arc and reduce muscle tone postoperatively, the anastomostic sacral nerve
root was only cut to match the diameter of intercostal-sural nerve,
corresponding to about 1/4-1/3 of the targeted nerve-root fascicula. An
inter-tract suture was made between the grafted sural nerve and the distal
nerve tract ends, two stitches being sufficient for connecting each nerve
bunch.
For patients who were subjected to
intradural anastomosis, because the grafted sural nerve segment is wider
than the nerve root (cauda equina) in the intradural space, the sural
nerve was divided into 2-3 fasciculus, each of which was anastomosed with
one nerve root. As such, one intercostal–sural nerve segment can be
connected to 2-3 nerve roots.
Finally, the connected nerves were bedded
within a thin layer of sacrospinalis muscle, and the vertebral canal was
covered with a muscle flap. Antibiotics and neurotrophic agents were
usually postoperatively administered, as well as hypertonic glucose and
potassium in the few patients who lost excessive cerebral spinal fluid.
Back bending was post-surgically restricted for four weeks
Assessments:
Urodynamic measurements included uroflow ratio, urethral pressure
distribution, CMG (cystometrogram) and cine-pressure-flow assessments.
Statistical significance was assessed using the t-student and X2
tests.
SLSEP (short-latency somatosensory evoked
potential) examination was performed using a Japanese-made MBE-2200
evoked-potential meter to record cauda equina (CE) electric potential and
N28 at 3 Hz stimulating frequency, 0.2 ms wave width, 5 ms/div scanning
rate, 20 UV input voltage and 10 Hz – 2 kHz frequency width. To assure
good repetition of different waves, the mean superposition was 1024
cycles.
RESULTS:
Thirty subjects were postoperatively
followed for 2-11 years with a 5-year average. Their outcomes are
summarized in the Table. Of the 30 subjects: 1) 26 (87%) recovered partial
defecation and urination sensation, 2) 23 (77%) regained the micturition
reflex and uriesthesis; 3) 19 (63%) recovered partial function of the
detrusor and sphincter urethra muscles; 4) 20 (67%) recovered partial
defecation function and sphincter contraction; and 5) 8 (27%) regained the
previous two functions (i.e., 3 & 4).
Defecation sensation appeared 3-16 months
(average 8) after the operation; the fecal and urinary reflex 7-24 months
(average 12); constriction of the detrusor and sphincter urethra muscles
24-48 months (average 28); and gluteal and perineal sensation 9-18 months
(average 12).
Electrophysiological assessments indicated
that the central nervous system had connected with the target system
through the transferred intercostal nerves. Specifically, the preoperative
wave width of CE and N28 was very low, and the latent period was either
prolonged or could not be elicited. After surgery, in 26 cases, the latent
period of CE was much shorter than the preoperative one. The wave
amplitude tended to be normal, and an N28, albeit abnormal, signal
appeared.
Table 1:
Comparison of Urodynamic Outcomes before & after Nerve Transfer
|
Parameters |
Cases
|
Preoperative |
Postoperative |
P value |
|
Maximum
Uroflow (ml/s) |
20 |
2.0+0.3 |
12.0+3.0 |
<0.05
|
|
Residual Urine
(ml) |
20 |
150+30 |
80+12 |
<0.05
|
|
Maximum
Bladder Volume (ml) |
30 |
150+30 |
200+30 |
>0.05 |
|
Maximum
constriction of detrusor muscle (cm H20) |
30 |
60+15 |
120+35 |
<0.05 |
|
Low Compliance
(N, %) |
30 |
26(87) |
9(30) |
<0.05
|
|
Absent
Detrusor Muscle Reflex (N, %) |
30 |
27(81) |
7(23) |
<0.05 |
|
Imbalance of
Detrusor & Extra-sphincter Muscles (N, %) |
30 |
5(17) |
4(13) |
>0.05 |
|
Intra-sphincter Muscle Spasms (N, %) |
30 |
10(33) |
3(10) |
>0.05 |
|
Extra-sphincter Denervation (N, %) |
30 |
7(23) |
2(7) |
>0.05 |
DISCUSSION:
Although the lower defecation and
urination center or reflex arc still exists in paraplegia above T12, these
functions are absent because the injury disrupts the connection to the
brain 2. As such, if a functional nerve-conduction pathway to
the brain can be re-established, some bowel and bladder function can be
restored. To restore this function, the reflex arc needs the assistance of
only a small amount of intact nerve fibers 3,4. The
nerve-bridging procedures described in this article used transferred
intercostal nerves to reach paralysis-affected nerves via the sacral nerve
roots and pelvic nerve plexus. As a result of re-establishing an intact
pathway from the brain to this plexus below the injury level, partial
bowel and bladder function was restored 5.
Although postoperative urodynamics and
extra-sphincter EMG confirmed new nerve connections 6,7,8,9,
restored bowel and bladder function does not solely derive from these
connections. Some patients can establish a so-called “trigger point” to
obtain urination reflex through percussing the muscle tendon 10.
Suturing the intercostal nerve or spinal nerve root with pelvic plexus or
sympathetic nerves can also restore this reflex. Although the success rate
by this alternative surgical approach was slightly higher than that
obtained through the percussion training, it was evidently lower than that
produced by the procedures discussed in this article. Furthermore, quality
of improvement was greater with the currently discussed procedures, which
also enhanced extra-sphincter muscle automatic sensation and constriction.
Postoperative functional training is
important, especially for patients who have depended on urethral
intubation. Patients were advised to do deep-breathing and breath-holding
training to establish urinary reflex because for the average person, both
intercostal-nerve-governed motor function and urination-and-defecation
activities involve coordinative muscular movements. It is quite natural
for patients who have recovered well after the operation to reconstruct
these functions. Concerning sphincter muscle constriction, patients who
lacked expert instructions or who did not persist with their training
often had poorer outcomes.
Postoperative outcomes were often less in
patients whose paraplegic level was below T12, who did not have
typical spastic paralysis, or who had traumatic or MRI-confirmed atrophic
changes in the medullary. Most of these patients suffered from urine
incontinence rather than retention, a situation in which the motor nerve
roots governing the sphincter muscle have became erosive. For these
patients, surgery should be performed within six months of injury, or,
alternatively, use the procedure in which the intercostal nerve is
connected to the pudendal nerve 11,12.
Finally, because benefits diminish with
increasing age, the procedure will be most suitable for patients younger
than 40.
CONCLUSION:
Restoration of bowel and bladder function
is a high priority for individuals with SCI and has been the focus of
other therapeutic interventions, such as functional electrical
stimulation. This specific surgery can consistently restore a significant
amount of such function. Although dramatic improvements have been observed
with the procedure, even modest benefits can have often have profound
quality-of-life implications by greatly increasing the personal
independence of individuals with SCI and, in turn, their societal
participation.
REFERENCES
|
1) Zhang S, Johnston L, Zhang Z, et
al. Restoration of Stepping-Forward and Ambulatory Function in
Patients with Paraplegia: Rerouting of Vascularized Intercostal Nerves
to Lumbar Nerve Roots using Interfascicular Anastomosis. Surg Technol
Int XI, 2003: 2003. |
|
2) Zhang S, Xiu X, Li Q, et al. Nerve
degeneration of lower extremity after paraplegia. J SMMU,
(editorial note: J. Second Military Medical University)1999; 20(3):
684-686. |
|
3) Pearson KG. Could enhanced reflex
function contribute to improving locomotion after spinal cord repair?
J Physiol, 2001; 533(Pt 1): 75-81. |
|
4) Yoshimura N. Bladder afferent
pathway and spinal cord injury: possible mechanisms inducing
hyperreflex of the urinary bladder. Prog Neurobiol, 1999,
57(6): 583-606. |
|
5) Sullivan. J. Spinal cord injury
research: review and synthesis. Crit Care Nurs Q, 1999; 22(2):
80-99. |
|
6) Prochazka, A, Mushahwar. VK. Spinal
cord function and rehabilitation - an Overview. J Physiol,
2001; 533(Pt 1): 3-4. |
|
7) Wang Z, Zhang S, and Cui Y. SLSEP
to evaluate 31 cases of vascularized intercostal nerve transfer in the
treatment of paraplegia. Ch J Plast Sur, 2000; 7(10): 94-6. |
|
8) Cui Y, Zhang, S, Zheng H, et al.
Diagnostic value of short latent period evoked potential in
vascularized intercostal nerve transfer in the treatment of
paraplegia. Clin J EEG, 2000; 2(2): 36-8. |
|
9) Zhang S, Qu C, Zhang X. An
urodynamic evaluation of neuroanastomosis for neurogenic bladder
caused by spinal cord injury. Ch J Urosurg, 2001; 22(4): 220-2. |
|
10) JI R, et al. Applied anatomy of
anastomosis of intercostal nerves to pudendal nerve for the treatment
of paraplegia. J Anat, 1989: 11(4): 291-294. |
|
11) Zhang, S, Zhang X, Ji, R. et al.
Functional reconstruction of peripheral nerves in paraplegia. J
Neurol Orthop Med Surg, 2000; 20(3): 89-97. |
TOP |
| |
| |
|
| |
| |
|
| |
| |
|
|